
Where are the transition metals on the periodic table? Metals are located on the left of the periodic table, and nonmetals are located on the upper right. Where are non-metals found on the periodic table? Reactivity increases as you move down the alkali metals group. The most reactive metals belong to the alkali metals element group. Which group contains the most reactive metals? Lithium, sodium, and potassium all react with water, for example. The elements toward the bottom left corner of the periodic table are the metals that are the most active in the sense of being the most reactive. Where are the reactive metals on the periodic table? All noble gases are colorless and odorless. As you can see in the periodic table in Figure below, noble gases include helium (He), neon (Ne), argon (Ar), krypton (Kr), xenon (Xe), and radon (Rn). Noble gases are nonreactive, nonmetallic elements in group 18 of the periodic table. What is nonreactive on the periodic table? The metals are to the left of the line (except for hydrogen, which is a nonmetal), the nonmetals are to the right of the line, and the elements immediately adjacent to the line are the metalloids. Where are the metals and non metals in the periodic table? By obtaining an electron, halogens form anions with a -1 charge. They have 7 electrons in their outermost shell and desire to gain an extra electron to complete their shell of 8 electrons. The halogens are so reactive due to their electronic configuration. Halogens are the most reactive nonmetals on the periodic table. What group contains the most nonreactive metals? The most nonmetallic elements(oxygen, fluorine, chlorine) occur at the top right of the Periodic Table. Metal characteristics tend to increase when moving from the top-right to bottom-left of the periodic table. Where are the most non reactive metals on the periodic table? 8 Where are the metalloids on the periodic table?. 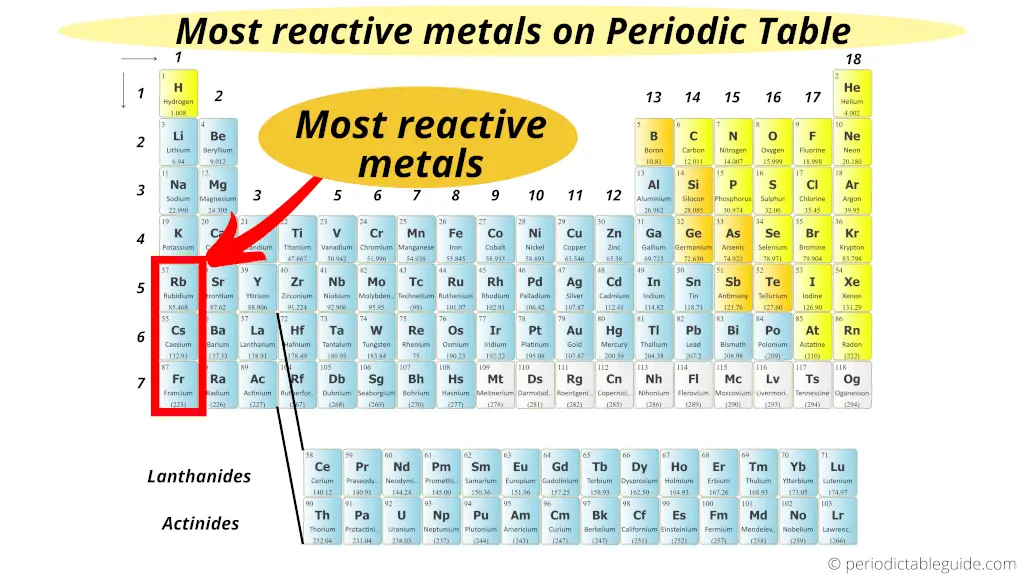
7 What kind of elements are on the left of the periodic table?.6 Which group on the periodic table is the least reactive?.5 Where are the transition metals on the periodic table?.4 Where are the reactive metals on the periodic table?.3 What is nonreactive on the periodic table?.2 What group contains the most nonreactive metals?.1 Where are the most non reactive metals on the periodic table?.These chemicals exist at all phases of matter including solid, liquid and gas they are the only elemental group that has this characteristic. The halogens also tend to be poor conductors of electricity and heat. Halogens are poisonous to humans on the whole, though each one is poisonous to a different degree. The further down the group you go, the less reactive the element is. The most reactive element from group seven is fluorine which is at the top of that section of the periodic table. The halogens include fluorine, chlorine, bromine, iodine and astatine. As a result, it often rips electrons away from other elements in order to gain this electron and become stable. Fluorine, for example, only has the choice of losing seven electrons or gaining one, so it almost always gains the one electron since this is much easier. They can gain electrons very quickly which is what makes them react so strongly to other elements. Halogens are highly reactive because they are all electronegative.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
